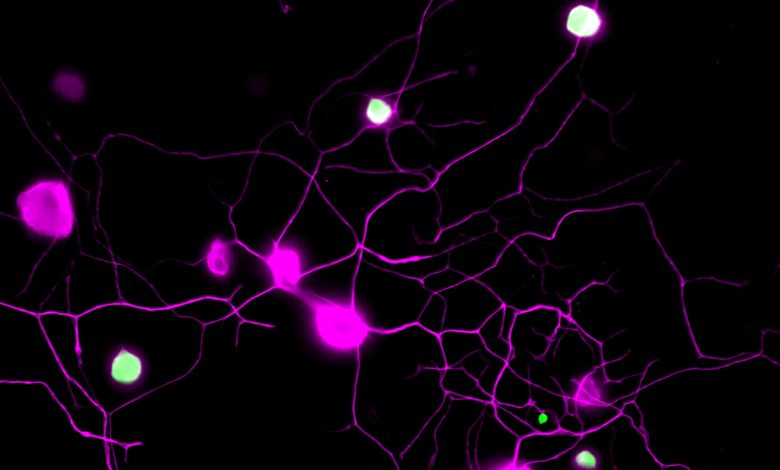
Lead Image: Mouse sensory neurons are shown in magenta. BH4, the molecule driving chronic pain, is shown in green. Hence, the neurons “in pain” are seen in green/white. Credit: ©Cronin/IMBA
Fresh hope for pain management from newly identified link between chronic pain and lung cancer in mice.
Although it is uncomfortable, pain is an important alarm system that alerts us to tissue damage and prompts us to withdraw from harmful situations. Although pain is expected to subside as injuries heal, many patients experience persistent pain long after recovery. Now, a new study points to possible new treatments for chronic pain with a surprising link to lung cancer. The work, which was published in Science Translational Medicine, was spearheaded by an international team of researchers at IMBA – Institute of Molecular Biotechnology of the Austrian Academy of Sciences, Harvard Medical School, and Boston Children’s Hospital. Their findings of the research, conducted in laboratory mouse models, open up multiple therapeutic opportunities that could allow the world to improve chronic pain management and curtail the opioid epidemic.
Acute pain is an important danger signal. On the other hand, chronic pain is based on persistent injury and can even be experienced in the absence of a stimulus, injury, or disease. Despite the fact that hundreds of millions of people are affected, chronic pain is among the least well-managed areas of healthcare. To improve how persistent pain is managed, especially in light of the raging opioid crisis, it is paramount to develop novel drugs based on a fundamental understanding of the underlying mechanisms.
“We had previously shown that sensory neurons produce a specific metabolite, BH4, which then drives chronic pain, such as neuropathic pain or inflammatory pain,” says project lead and co-corresponding author Shane Cronin. He is a staff scientist in the Penninger lab at IMBA and a former postdoc in the Woolf lab at Harvard Medical School and F.M. Kirby Neurobiology Center, Boston Children’s Hospital. “The concentrations of BH4 correlated very well with the pain intensity. So, we naturally thought that this was a great pathway to target.”
To identify drugs that reduce BH4 levels in pain neurons, the scientists performed a “phenotypic screen” of 1000 target-annotated, FDA-approved medications. This approach allowed the researchers to start their search using medications that are currently in use for various indications, and to identify undescribed, off-target analgesic properties. Among the initial findings of this hypothesis-driven search, the team was able to link the previously observed analgesic effects of several drugs, including clonidine and capsaicin, to the BH4 pathway. (Clonidine is a prescription medication typically used to lower blood pressure and capsaicin is the active component of chili peppers.)
“However, our phenotypic screen also allowed us to ‘repurpose’ a surprising drug,” says Cronin. The drug ‘fluphenazine’, which is an antipsychotic that has been used to treat schizophrenia. “We found that fluphenazine blocks the BH4 pathway in injured nerves. We also demonstrated its effects in chronic pain following nerve injury in vivo.” The scientists also found that the effective analgesic dose of fluphenazine in their experiments in the mouse model is comparable to the low end of the doses safely indicated for schizophrenia in humans.
Additionally, the screen uncovered a novel and unexpected molecular link between the BH4 pathway and EGFR/KRAS signaling, a pathway involved in multiple cancers. Blocking EGFR/KRAS signaling reduced pain sensitivity by decreasing the levels of BH4. The genes of EGFR and KRAS are the two most frequently mutated genes in lung cancer, which prompted the researchers to look at BH4 in lung cancer. Surprisingly, by deleting an important enzyme, GCH1, in the BH4 pathway, the mouse models of KRAS-driven lung cancer developed fewer tumors and survived much longer. Hence, the research team uncovered a common signaling pathway for chronic pain and lung cancer through EGFR/KRAS and BH4, thus opening up new avenues of treatment for both conditions.
“Chronic pain is currently subjected to often ineffective palliative treatments. Furthermore, effective painkillers such as opioids can lead, if used inappropriately, to severe addiction. It is therefore critical to find and develop new and repurposed drugs to treat chronic pain,” says co-corresponding author Clifford Woolf, professor of neurology and neurobiology at Harvard Medical School and director of the F.M. Kirby Neurobiology Center at Boston Children’s Hospital.
One intriguing aspect of the study is the mechanistic link between pain and lung cancer. “The same triggers that drive tumor growth appear to be also involved in setting the path to chronic pain, often experienced by cancer patients. We also know that sensory nerves can drive cancer, which could explain the vicious circuit of cancer and pain,” adds co-corresponding author Josef Penninger. He is an IMBA group leader and founding director, who is currently also the director of the Life Sciences Institute at the University of British Columbia (UBC), Vancouver, Canada. “Understanding these cross-talks is therefore not only critical for cancer treatments but might also help to improve the quality of life for cancer patients towards less pain.”
Reference: “Phenotypic drug screen uncovers the metabolic GCH1/BH4 pathway as key regulator of EGFR/KRAS-mediated neuropathic pain and lung cancer” by Cronin, S. J. F., et al., 31 August 2022, Science Translational Medicine.
DOI: 10.1126/scitranslmed.abj1531
Funding: Harvard Medical School, Austrian Federal Ministry of Education, Science and Research, Austrian Academy of Sciences, City of Vienna, Austrian Science Fund, T. von Zastrow foundation, a Canada 150 Research Chairs Program, Canadian Institutes of Health Research, Harvard Stem Cell Institute, NIH/National Institutes of Health