The stuff we see in science fiction movies has actually come to life.
3D printing living tissue—including corneas, blood vessels and skin—is no easy task. But at least it’s all living tissue. Bone, by contrast, is a mixture of living and inorganic compounds in a highly structured mineral matrix.
3D printing bone, in other words, is a challenge within a challenge.
Which is why bioengineers have tried so many different materials for their synthetic bones—including hydrogels, thermoplastics, and bioceramics. Now, a team at the University of New South Wales in Sydney, Australia, has developed a ceramic ink that can be 3D-printed at room temperature with live cells and without harsh chemicals—a notable improvement over earlier technologies. The new technique could eventually be used to print bone directly into a patient’s body, the researchers say.
A new innovation allows scientists to 3D-print human bones from a person’s own living cells and for the first time, the process has been done at room temperature.
A team at Australia’s University of New South Wales-Sydney created a ‘bio-ink’ gel that contains a patient’s live bone cells in a calcium phosphate solution, which are necessary minerals for bone formation and maintenance.
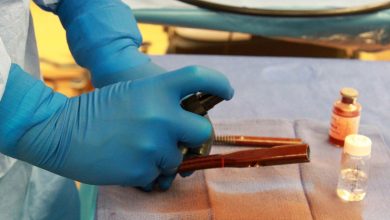
Using a technique known as ceramic omnidirectional bioprinting in cell-suspensions (COBICS), the gel is 3D-printed directly into the patient’s bone cavity instead of surgeons having to remove a piece from a different location.
The material then hardens within minutes of being exposed to bodily fluids and converts into mechanically interlocking bone nanocrystals.
The act of 3D-printing bone-mimicking structures is not new, but the University of New South Wales-Sydney’s method allows the process to be done at room temperature for the first time.
This means bones can be created on the spot inside a medical room, along with using the patient’s own living cells.
Dr Iman Roohani from UNSW’s School of Chemistry, said: ‘This is a unique technology that can produce structures that closely mimic bone tissue.’
“In contrast to previous materials, our technique offers a way to print constructs in situ which mimic the structure and chemistry of the bone,” stated Iman Roohani. “The opportunities are limitless.”
‘It could be used in clinical applications where there is a large demand for in situ repair of bone defects such as those caused by trauma, cancer, or where a big chunk of tissue is resected.’
Prior to this work, if a patient needed a piece of bone doctors would have to remove a section from a different location in the body.

And 3D-printing was only available by first going to a laboratory to fabricate the structures using high-temperature furnaces and toxic chemicals.
Associate Professor Kristopher Kilian who co-developed the breakthrough technology, said: ‘This produces a dry material that is then brought into a clinical setting or in a laboratory, where they wash it profusely and then add living cells to it,’ Professor Kilian says.
‘The cool thing about our technique is you can just extrude it directly into a place where there are cells, like a cavity in a patient’s bone. We can go directly into the bone where there are cells, blood vessels and fat, and print a bone-like structure that already contains living cells, right in that area.’
‘There are currently no technologies that can do that directly.’
The special ink made for the process forms a structure that is chemically similar to bone-building blocks, according to the scientists.
‘The ink is formulated in such a way that the conversion is quick, non-toxic in a biological environment and it only initiates when ink is exposed to the body fluids, providing an ample working time for the end-user, for example, surgeons,’ Dr Roohani said.
He explains that the ink combines with a collagenous substance that contains living cells, ‘it enables in-situ fabrication of bone-like tissues which may be suitable for bone tissue engineering applications, disease modelling, drug screening, and in-situ reconstruction of bone and osteochondral defects.’
3D-printed bone tissue has plentiful medical and research applications: modelling bone disease; drug screening; studying bone’s unique microenvironment; and perhaps most notably, repairing damaged bone in cases of trauma, cancer or other illnesses. The current gold standard for repairing bone is an autologous bone graft—harvesting bone from another part of the patient’s own body. Unfortunately, autologous grafts are associated with high rates of infection and don’t work if the needed amount of bone material is too large.
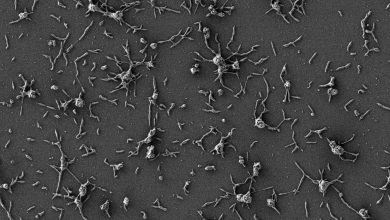
In an effort to create a synthetic bone material as similar to an autologous graft as possible, Roohani, biochemist Kristopher Kilian and colleagues at UNSW made an ink that could be 3D printed into an aqueous environment like the body. After two years of refining, they created a biocompatible calcium phosphate material that forms a paste at room temperature. When put into a gelatin bath or other solution, a chemical reaction occurs and the paste hardens into a porous nanocrystal matrix similar to structure of native bone tissue.
The work is described this week in the journal Advanced Functional Materials.