
Lead Image: Salk Institute researchers have developed a new compound that acts like a master reset switch in the intestines. In a new study, the compound, called FexD, is found to prevent and reverse intestinal inflammation in mouse models of inflammatory bowel disease.
New therapeutic has the potential to treat inflammatory bowel disease by targeting a molecule that keeps order in the intestines.
Salk Institute scientists have developed a new drug that acts like a master reset switch in the gut. Called FexD, the compound has previously been found to burn fat, lower cholesterol, and ward off colorectal cancer in mice. Now, the team reports that FexD can also prevent and reverse intestinal inflammation in mouse models of inflammatory bowel disease. The study was published on December 12, 2022, in the journal Proceedings of the National Academy of Sciences.
“The Salk-developed drug FexD provides a new way to restore balance to the digestive system and treat inflammatory diseases that are currently very difficult to manage,” says Salk Professor Ronald Evans, senior author of the study. Evans is also director of Salk’s Gene Expression Laboratory and March of Dimes Chair in Molecular and Developmental Biology.
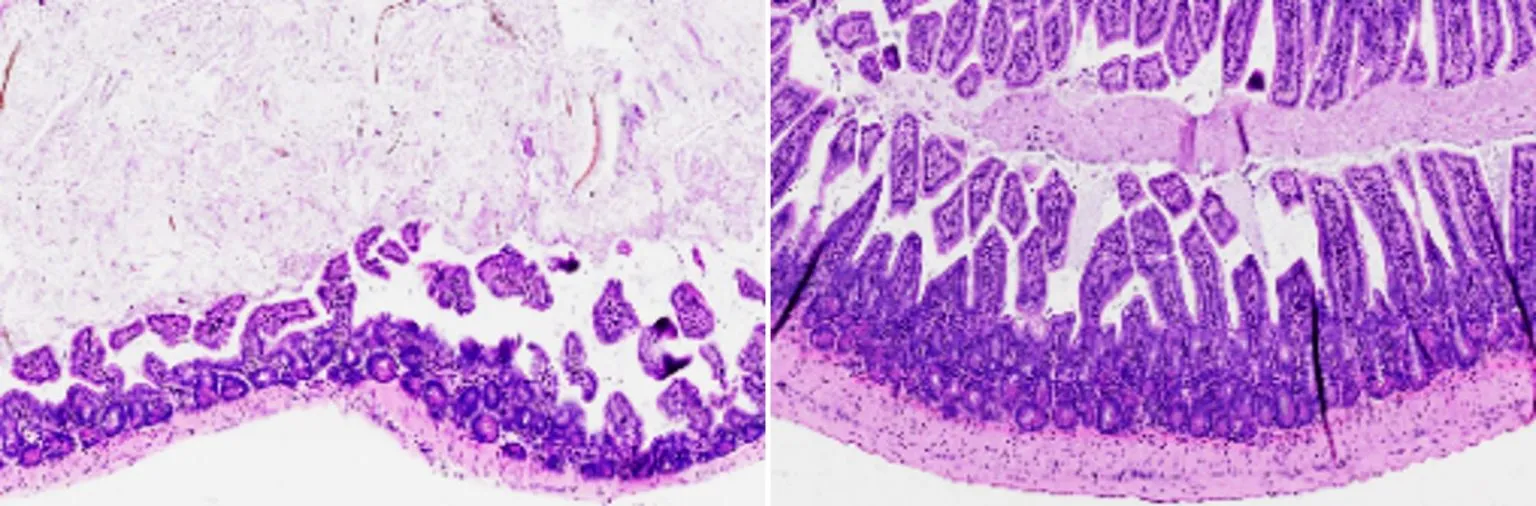
Inflammatory bowel disease (IBD), which includes both Crohn’s disease and ulcerative colitis, is estimated by the CDC to affect approximately 1.3% of American adults (about 3 million people). It is a condition that is characterized by chronic inflammation of the gastrointestinal tract with an excess of immune cells and inflammatory signaling molecules known as cytokines in the gut. Existing treatments mostly work by either suppressing the entire immune system or by targeting individual cytokines. They are only effective for some patients and carry a large number of significant side effects.
Evans’ lab has studied Farnesoid X receptor (FXR) for more than two decades. FXR is a master regulator protein that senses the bile acids delivered to the digestive system to help digest food and absorb nutrients. When FXR detects a shift in bile acids at the beginning of a meal, it prepares the body for an influx of food by flipping on and off dozens of cellular programs related to digestion, blood sugar, and fat metabolism.

Evans and his colleagues developed a pill called fexaramine in 2015 that activates FXR in the gut. They initially showed the pill can stop weight gain and control blood sugar in mice. In 2019, they showed that an updated version of fexaramine—FexDan—also prevented cancer-associated changes to stem cells in the gut. Their work suggested that FXR also played a role in regulating inflammation.
“Every time you eat, you’re causing small amounts of inflammation in your gut as your intestinal cells encounter new molecules. FXR makes sure inflammation stays under control during normal feeding,” says Senior Staff Scientist Michael Downes, co-corresponding author of the new paper.

In the new work, Evans’ group discovered that activating FXR can be used to ease symptoms in inflammation-driven diseases. When the researchers gave mice with IBD a daily dose of oral FexD, either before or after the onset of intestinal inflammation, the drug prevented or treated the inflammation. By activating FXR, FexD reduced the infiltration of a class of highly inflammatory immune cells called innate lymphoid cells. In turn, levels of cytokines already implicated in IBD decreased to levels normally seen in healthy mice.
“When we activate FXR, we restore appropriate signaling pathways in the gut, bringing things back to a homeostatic level,” says Senior Research Scientist Annette Atkins, co-author of the study.
Cytokines are not completely blocked by FexD because FXR acts more like a reset button than an off switch for the immune system. This means that after a dose of FexD, the immune system continues functioning in a normal way. The compound still must be optimized for use in humans and tested in clinical trials, but the researchers say their findings provide important information about the complex links between gut health and inflammation and could eventually lead to an IBD therapeutic.
“In people with IBD, our strategy could potentially be very effective at preventing flare-ups and as a long-term maintenance drug,” says first author Ting Fu, previously a postdoctoral fellow at Salk and now an assistant professor at the University of Wisconsin-Madison.
Reference: “FXR mediates ILC-intrinsic responses to intestinal inflammation” by Ting Fu, Yuwenbin Li, Tae Gyu Oh, Fritz Cayabyab, Nanhai He, Qin Tang, Sally Coulter, Morgan Truitt, Paul Medina, Mingxiao He, Ruth T. Yu, Annette Atkins, Ye Zheng, Christopher Liddle, Michael Downes and Ronald M. Evans, 12 December 2022, Proceedings of the National Academy of Sciences.
DOI: 10.1073/pnas.2213041119
Other authors of the paper include Yuwenbin Li, Tae Gyu Oh, Fritz Cayabyab, Nanhai He, Qin Tang, Morgan Truitt, Paul Medina, Mingxiao He, Ruth T. Yu, and Ye Zheng of Salk; and Sally Coulter and Christopher Liddle of the University of Sydney.
The work was supported in part by the National Institutes of Health (DK057978, HL105278, and HL088093), the National Cancer Institute (CA014195), the National Health and Medical Research Council of Australia (grant 1087297) the Leona M. and Harry B. Helmsley Charitable Trust (2017PG-MED001), an SWCRF Investigator Award, a Hewitt Medical Foundation Fellowship, a Salk Alumni Fellowship, a Crohn’s & Colitis Foundation (CCFA) Visiting IBD Research Fellowship, a Stand Up To Cancer-Cancer Research UK-Lustgarten Foundation Pancreatic Cancer Dream Team Research Grant (SU2C-AACR-DT-20-16), the Howard Hughes Medical Institute, the NOMIS Foundation, and the National Institute of Environmental Health Sciences (P42ES010337).





