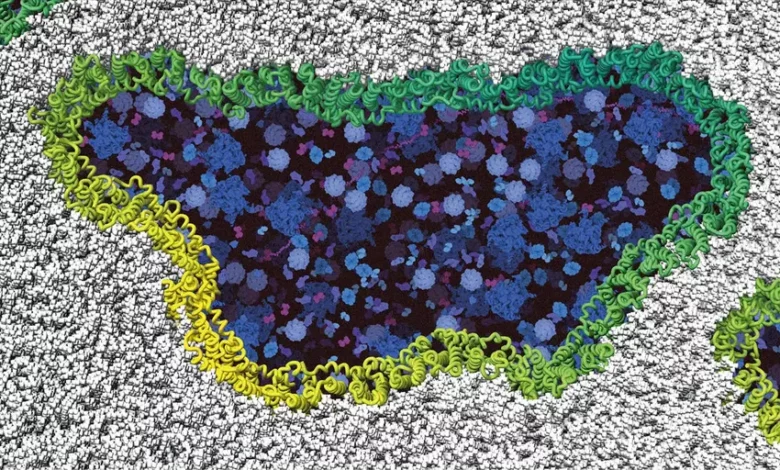
Ninjurin-1 proteins assemble (green/yellow) into filaments and rupture the cell membrane (gray) until the cell disintegrates completely. Intracellular components are shown in blue. Credit: Biozentrum, University of Basel
In our bodies, millions of cells meet their end on a daily basis. Contrary to popular belief, cells don’t just explode when they die. Instead, a particular protein acts as a trigger for the rupture of the cell membrane. Scientists from the University of Basel have recently been able to elucidate the exact mechanism at the atomic level. Their findings are published in the journal Nature.
The self-elimination of cells is a vital process for all living organisms. When cells become damaged or infected with viruses or bacteria, they initiate an internal “self-destruct” sequence. This essential mechanism wards off the potential growth of tumors and prevents the spread of harmful pathogens throughout the body.
Until recently, it was assumed that cells simply burst and die at the end of their life. Now, researchers at the Biozentrum of the University of Basel, the University of Lausanne, and the Department of Biosystems Science and Engineering (D-BSSE) at ETH Zurich have provided new insights into the final step of cell death.
In the scientific journal Nature, they describe how a protein called ninjurin-1 assembles into filaments that work like a zipper and open the cell membrane, thus leading to the disintegration of the cell. The new insights are an important milestone in the understanding of cell death.
Protein acts as a breaking point in the cell membrane
Various signals, such as bacterial components, trigger the cell death machinery. At the final stage of this process, the cell’s protective membrane is compromised by tiny pores which allow ions to stream into the cell.
“The common understanding was that the cell then swells until it finally bursts due to increasing osmotic pressure,” explains Professor Sebastian Hiller who heads a research group at the Biozentrum, University of Basel. “We are now resolving how the cells really rupture. Instead of bursting like a balloon, the protein ninjurin-1 provides a breaking point in the cell membrane, causing rupture at specific sites.”
At the end of their lives, cells do not simply burst. Instead, a specific protein serves as a breaking point for the cell membrane rupture. SNI PhD student Morris Degen (Biozentrum, University of Basel) explains how this mechanism works. Credit:
Swiss Nanoscience Institute
Using advanced techniques such as highly sensitive microscopes and NMR spectroscopy, the scientists have been able to elucidate the mechanism by which ninjurin-1 induces membrane rupture at the level of individual atoms. Ninjurin-1 is a small protein embedded in the cell membrane.
“Upon receiving the suicide command, two ninjurin-1 proteins initially cluster together and drive a wedge into the membrane,” explains Morris Degen, first author of the study and Ph.D. student at the Ph.D. School of the Swiss Nanoscience Institute. “Large lesions and holes are formed by many further proteins attaching to the initial wedge. In this way, the cell membrane is cleaved open piece by piece until the cell disintegrates completely.”
The cell debris is then removed by the body’s own cleaning service.
“It is now evident that the cells do not burst without ninjurin-1. They do swell to a certain extent due to the influx of ions, but membrane rupture is contingent on the function of this protein,” adds Hiller. “The textbook’s chapter on cell death will be expanded with these beautiful structural insights.”
Therapy to prevent or promote cell death
A deeper understanding of cell death will facilitate the search for novel drug targets. Therapeutic interventions to treat cancer are conceivable since some tumor cells evade programmed cell death.
Also, in the case of premature cell death observed in neurodegenerative diseases such as Parkinson’s disease or in life-threatening conditions such as septic shock, drugs that interfere in this process are a potential treatment option.
Reference: “Structural basis of NINJ1-mediated plasma membrane rupture in cell death” by Morris Degen, José Carlos Santos, Kristyna Pluhackova, Gonzalo Cebrero, Saray Ramos, Gytis Jankevicius, Ella Hartenian, Undina Guillerm, Stefania A. Mari, Bastian Kohl, Daniel J. Müller, Paul Schanda, Timm Maier, Camilo Perez, Christian Sieben, Petr Broz and Sebastian Hiller, 17 May 2023, Nature.
DOI: 10.1038/s41586-023-05991-z



